| Outline |
|---|
|
In the eukaryote, genomic DNA is packaged into chromatin that appears to affect all stages of transcription, including initiation and elongation.
Eukaryotic gene expression is regulated by chromatin structure together with the cellular network of cis-acting elements and trans-acting factors.
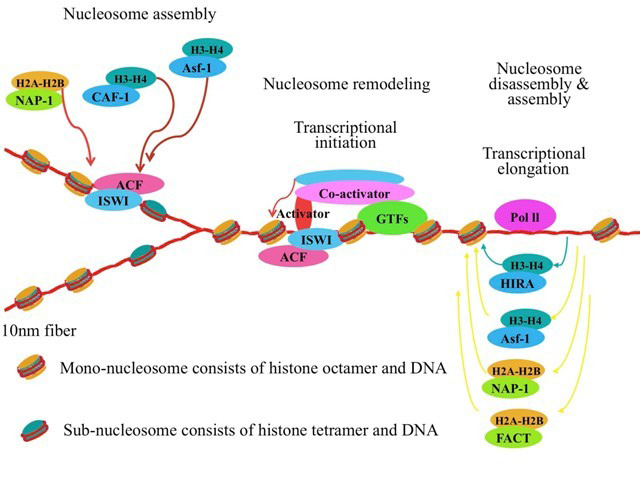
The nucleosome is the fundamental unit of chromatin and is composed of147 base pairs (bp) of DNA wrapped 1.65 turns around the histone octamer of the four core histones (H2A, H2B, H3, and H4) (for review see 1).
According to recent concepts of the histone modification network, post-translational modifications of the histone tail control the structure and function of chromatin including replication, transcription and repair.
It is known that dis-regulation of chromatin modification cause malignancies. However, it is not proven that the change of histone modification by itself is the initial causal event of carcinogenesis. Recently, we found that histone modification by itself can be the causal event of carcinogenesis. Briefly histone H2A Thr 120 hyper-phosphorylation by hVRK1 causes inappropriate gene expression including up-regulated cyclin D1, resulting in carcinogenesis2. 1 Higashi, M., Inoue, S. & Ito, T. Core histone H2A ubiquitylation and transcriptional regulation. Exp Cell Res 316, 2707-2712, (2010). 2 Aihara, H. et al. Histone H2A T120 Phosphorylation Promotes Oncogenic Transformation via Upregulation of Cyclin D1. Mol Cell 64, 176-188, (2016). |